More On:
COVID vaccine
‘I can’t find my mask!’: Biden frantic after forgetting he put it in his pocket
People are having vaccination parties to celebrate the end of lockdown
As virus engulfs India, diaspora watches with despair
Joe Rogan walks back anti-vaxx comments: ‘I’m not a doctor, I’m a f–ing moron’
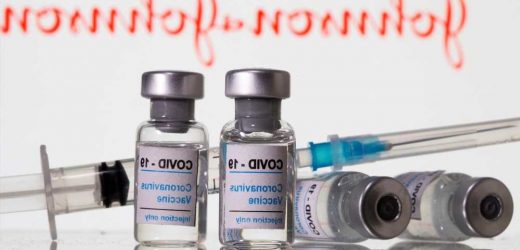
Anxiety triggered adverse reactions to the Johnson & Johnson COVID-19 vaccine in at least 64 people — including cases that resulted in jab sites being shut down, the Centers for Disease Control and Prevention said Friday.
The CDC studied the physical reactions, including dizziness and fainting, in dozens of people who received the shots in five states between April 7 and 9, officials said.
Many of the people reported nausea, vomiting and chest pain in California, Colorado, Georgia, Iowa and North Carolina. Others said they had an elevated heart rate.
The reactions prompted some vaccine centers to temporarily shut down while health officials investigated. But the CDC determined those reactions were brought on by anxiety caused by receiving injections.
“We knew we were going to see this,” said Dr. Noni MacDonald, a Canadian researcher who has studied similar incidents.
“Of the three COVID-19 vaccines authorized in the U.S., only J&J requires just one dose. That probably makes it more appealing to people who are nervous about shots and might leave them highly predisposed to anxiety-related events,” the CDC report states.
Some of the vaccine centers under review advertised that they were giving Johnson & Johnson shots, said Dr. Tom Shimabukuro, who leads the CDC’s COVID-19 vaccine safety monitoring work and is one of the study’s authors.
The CDC also found that about one-quarter of the people reporting side effects had similar reactions following past vaccinations.
Some experts have referred to the phenomenon as a form of mass hysteria, but MacDonald rejected the notion.
“These people are not crazy” — just feeling physical responses to psychological stress, she said.
Many people who experience stress-related symptoms are younger, and past cases have involved students. Some hyperventilate and report headaches, which may at first appear to be more severe, neurological symptoms, she said.
The adverse reactions in the study are not related to the rare blood-clotting condition that led federal health officials to put the brakes on the Johnson &Johnson vaccine earlier this month.
With Post wires
Share this article:
Source: Read Full Article