A fungal pathogen wreaks havoc, even turning people into zombies – which is so far-fetched, right? Well, not entirely.
The year is 2023. The world is gripped by a pandemic. A small group of people weave their way through a ruined city. They step into an abandoned building, hear a noise – a strange, inhuman clicking. Instantly, they know they are near a person infected with the plague that has killed their world – a fungus that takes over the brain of its human hosts, turning them into shambling monsters. This creature lurches past them, cauliflower-like growths over its eyes – but its ears work very well. One of the healthy humans steps on broken glass. The monster shrieks – and leaps at them.
That is a typical scene from the hit TV show The Last of Us, which recently started airing to instant critical and ratings success. Based on the award-winning 2013 video game of the same name, the show is set in an alternate world in which modern civilisation as we know it has crumbled and humanity is divided into walled-off quarantine zones governed by martial law in an effort to stop the spread of the plague.
The TV show is a dark imagining of how humanity would deal with such a crisis but, surely, its central premise – that a fungal pathogen can rapidly cause the collapse of civilisation – is pure science fiction? Right? Well, not entirely. The fungus that causes the plague in the show and game, a type of Cordyceps, really exists. In fact, it was an inspiration for the writer of The Last of Us. And fungal epidemics are a thing too. Real-life experts are very worried about them.
Can a fungus harm humans en masse? Can it even turn us into zombies? And what do we have to fight back with?
The heroes of The Last of Us. Credit:
What’s a fungus?
Fungi are everywhere in the world, in the air, in the soil and in our bodies. Scientists used to put them in the same class as plants, but their complexity and strangeness made them a poor fit – so now they get to be their own thing. There are about 144,000 known species.
They are culinarily important; we eat mushrooms and truffles, and use yeast to make bread, wine, beer and cheese. In humans, they help and harm. Fungi make up an important component of your microbiome; you have fungi happily living all around your body. Penicillin and many other antibiotics are manufactured by fungi. Some species also cause tinea and athlete’s foot and thrush, unpleasant but not life-threatening. That annoying-but-harmless reputation contributed to complacency about fungi for a long time, say researchers who study them. Big mistake. Because serious fungal infections are now taking off around the world.
“It is a bit daunting … This is not your simple toenail infection. It’s really scary stuff.”
“It is a bit daunting. It’s multi-drug resistant and we’re really running out of options for treatment. This is not your simple toenail infection. It’s really scary stuff,” says Dr Claudia Simm, a fungus researcher at Monash University.
Rather than being a simple-celled prokaryote as with bacteria, a fungus is a eukaryote – it has complex cells, just like humans. That gives it access to a range of unique tricks and, worse, means it is hard to fight with drugs, as things that might kill it also tend to kill human cells (more on this in a moment).
In The Last of Us, the infection is spread by zombies biting healthy people, or it is breathed in. In real life, fungi are spread via airborne spores, but they do not spread from person to person, “which is why they are not going to cause a pandemic”, says Professor Ana Traven, co-director of an emerging superbug threats training program at Monash University.
For most healthy people, the fungal spores in the air are not a threat. We breathe them in and out every day, but they typically don’t make healthy people sick, as our immune systems are well capable of killing them off. Healthy people do get fungal infections – Coccidioidomycosis, or valley fever, is one example – but in general life-threatening fungal infections are rare.
The people who are really threatened are the immunocompromised, which is why virtually all life-threatening fungal infections happen in hospitals.
A poster from The Last of Us.
OK, so a fungus can’t turn people into zombies?
Uh, no. “There is definitely no fungus that would do something like this to a human,” says Traven. “But it does happen in the insect world.”
The Last of Us focuses on Cordyceps, a genus that includes a number of species of fungi. But the real-life inspiration is actually a specific fungal villain once known as Cordyceps unilateralis (now called Ophiocordyceps unilateralis after a scientific name change) which really does turn its prey into zombies. The tropical-forest fungus infects ants via spores and takes control. The ant is prompted to leave its nest, heading for more humid areas preferred by the fungus. The fungus drives the ant downward until sitting on a leaf about 10 inches off the ground; the ant then bites hard into a leaf and “waits for death”, according to a National Geographic report. When it’s ready, the fungus pushes a fruiting body up through the dead ant’s head which explodes into spores, spreading the fungus to other victims.
A moth is taken over by a Cordyceps fungus in the Ecuadorian Amazon.Credit:Getty Images
Unilateralis isn’t the only zombie fungus out there. Entomophthora muscae preys on houseflies. The fungus gets into the fly’s brain, forcing the fly to crawl upwards to the top of a branch. It then drools a glue-like substance, likely manufactured by the fungus, that sticks it hard in place. The fungus finishes off eating the fly, covering its body in puffy white filaments.
Other houseflies seem to be strongly attracted to their zombie colleague, inspecting, touching and even trying to copulate with the poor creature, for reasons scientists still don’t understand. This is exactly what the fungus wants. The fungus grows tiny stalks, which look like miniature cannons, through the housefly’s body; when set off by motion from nearby houseflies, these cannons explode, spraying spores onto the next victim.
But these two examples are not just weird anomalies; they are the tip of a fungal iceberg. Fungi that make their hosts light-sensitive or hyperactive have been found. Massospora cicadina turns cicadas into “horror-movie sex bots”, in the words of Scientific American. The fungus is sexually transmitted, and even prompts male cicadas to act like females – allowing them to spread the fungus to unsuspecting male colleagues.
The fungus grows tiny stalks, which look like miniature cannons, through the housefly’s body; when set off by motion, these cannons explode.
How do these fungi manage to manipulate insect behaviour so profoundly? One theory: drugs. Massospora may produce its own cathinone, an amphetamine stimulant found in illegal drugs; other samples of the same fungus have shown evidence for psilocybin, the key ingredient in magic mushrooms.
Other species of fungus directly infect the brain, possibly directly manipulating behaviour; still others use very specific behaviour-manipulating toxins.
A Cordyceps fungus in a stick insect in a tropical rainforest.Credit:Adobe Stock
So, we’re not going to become zombies, but we should still be concerned?
Well, yes. Fungi are not going to cause a global pandemic, but they are a growing threat worldwide. There are about 1.7 billion fungal infections a year. Candida alone, one of the fungi that scientists are most worried about, causes about 400,000 bloodstream infections every year, with a mortality rate exceeding 40 per cent. There are not many statistics about this from Australia. In one study, researchers looking at data from four cancer and stem cell wards at Australian children’s hospitals between 2004 and 2013 found 337 records of fungus infections, with a mortality rate between 4 and 28 per cent; 56 children died.
In an effort to draw attention – and funding – to the fungus threat, the World Health Organisation (WHO) in 2022 named a big four of fungal “priority pathogens”, a rogues’ gallery of fungi that present the greatest public health threat.
Candida auris … has been labelled the new “fungal superbug” because of its resistance and the fact that many of the people it infects have just undergone surgery.
There’s Cryptococcus neoformans, a soil yeast that can be inhaled by humans, where it spreads from the lungs to the blood and brain. It preys on the immunocompromised, particularly those with HIV; mortality is between 41 and 61 per cent.
Candida auris is a type of yeast that can infect the blood, bones and organs and which has an extremely high mortality rate of between 29 and 53 per cent. The fungus has been responsible for several hospital outbreaks, with infection trends increasing across the world; it is resistant to many of the antifungal medications we have. This fungus has been labelled the new “fungal superbug” because of its resistance and the fact that many of the people it infects have just undergone surgery.
Aspergillus fumigatus is a mould that can infect the lungs, nervous system and other organs. Mortality rates from antifungal-resistant specimens are up to 100 per cent in some studies; it too preys on the immunocompromised. Infection rates are increasing globally.
Candida albicans is a yeast that makes up part of our human microbiome, particularly our mouth, gut and skin, and is generally harmless. But if it becomes invasive (say, entering the bloodstream) it has a mortality rate of between 20 and 50 per cent.
Those are just the big four. The WHO highlighted another 15 that pose a grave threat. “Emerging from the shadows of the bacterial antimicrobial resistance pandemic, fungal infections are growing, and are ever more resistant to treatments, becoming a public health concern worldwide,” says Dr Hanan Balkhy, the WHO’s assistant director-general for antimicrobial resistance.
Unlike bacteria, for which we have a growing arsenal of antibiotics, we have very few weapons to fight fungus.
Justin Beardsley is an associate professor in Infectious Diseases at the University of Sydney with a special interest in fungal pathogens, and was one of the researchers who contributed to the WHO report. “There are big threats to humanity from fungal pathogens – not in a zombie apocalypse way, but in terms of untreatable infections,” he says.
Unlike bacteria, for which we have a growing arsenal of antibiotics, we have very few weapons to fight fungus. There are just four classes of medicine available, far fewer than we have antibiotics and far fewer than we need (to put that in context, US regulators approved nine new antibiotics in 2018-19 alone). Few anti-fungal medicines are in the research pipeline.
All of these medicines are also fairly rough on the patient because of an eerie complication – in terms of their cellular makeup, fungi are closer to humans than they are to plants. This means that a lot of the drugs that target their cells are also toxic to ours. “Some of the drugs which we have to combat fungal infections were originally being tested as potential chemotherapy drugs,” Beardsley says. “They weren’t suitable for that, but it was found they were effective against fungal cells.”
Many fungi already have acquired resistance to our anti-fungus medications. This is likely being driven by the heavy use of anti-fungal pesticides in farming; the pesticides end up in the soil, where they drive fungi to evolve around them. Some samples of Aspergillus fumigatus from Asia have been found to be more than 80 per cent resistant to anti-fungal sprays.
Bella Ramsey (left) and Anna Torv in The Last of Us.Credit:HBO/Binge
What does climate change have to do with it?
It might be key to the whole story. Get this: until 2009 no one, anywhere in the world, had ever heard of Candida auris. Then, out of nowhere, it emerged in multiple hospitals in different countries, from Japan to Venezuela, at the same time, menacing medicos and killing patients (and, likely thanks to the farming problem, it’s already resistant to all our anti-fungal drugs).
Auris is the standout, but we see the same problem across the board: in the past 20 years, the world has seen a major spike in life-threatening fungal infections. Scientists don’t know why, but suspicions centre on the climate crisis. Fungi, on the whole, actually do not love infecting mammals. Our body temperatures are slightly higher than fungi find comfortable.
“The reason we’re seeing these outbreaks of fungal infection that we did not see 20 years ago is really because of global heating.”
In the first episode of The Last of Us, a scientist on a talk show points out that if the climate were to warm by just a few degrees, fungi might adapt to tolerate higher temperatures, which would make it much more amenable to the human body as a host. Beardsley says there is some truth to that assertion. “There’s even suggestions that one of the reasons mammals have such high body temperatures is because of the presence of fungi, it became (evolutionarily) beneficial to have a high body temperature.”
But the temperature gap keeping us safe from fungi is small, just a few degrees. And the world has already warmed 1.5 degrees since pre-industrial times. As fungi mutate to tolerate higher heat levels, a range of new environments open up for them, including human beings, who are full of the sugars they love to consume. “The reason we’re seeing these outbreaks of fungal infection that we did not see 20 years ago is really because of global heating,” says Traven.
And this problem is likely to get worse, with many more fungal pathogens in the environment than those known to infect humans.
“If the difference between the ambient temperature of the environment and human body temperature becomes less, then there’s millions of fungal pathogens which will have to adapt. So there’s a theoretical risk there,” says Beardsley.
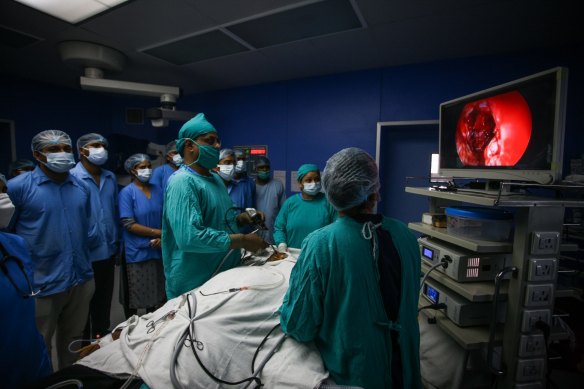
A specialist removes mucormycosis from a recovering COVID patient in Allahabad, India. Credit:Getty Images
What effect is COVID having?
The recent pandemic had the effect of suddenly inflating the number of people with weakened or compromised immune systems around the world, and fungi were ready to take advantage of the situation.
Beardsley points to one example recorded in India during the pandemic in patients who had COVID-19 as well as diabetes who were given steroids as a way to head off the worst effects of their viral infection.
Changing global health landscapes, such as a viral pandemic, could have a knock-on effect and trigger a fungal epidemic.
That combination made them much more susceptible to mucormycosis, a rare but serious fungal infection. The fungus is found in the soil, plants and decaying food – and our immune system is normally perfectly capable of dealing with it. India reported more than 8800 cases of the mould, which has a 50 per cent mortality rate; the country’s health ministry called for the formal declaration of a fungal epidemic.
“They weren’t dying from COVID, they were dying from this fungal infection in their sinuses that eats into the brain,” says Beardsley. “That was very eye-opening – that these sorts of fungal infections could complicate a viral respiratory pandemic.”
These are the sort of circumstances that could possibly give rise to a fungal epidemic, says Traven. “Changing global health landscapes, such as a viral pandemic (which will unfortunately happen again), could have a knock-on effect and trigger a fungal epidemic. This is what happened in India.”
What is being done about these fungal infections?
Research funding is not anywhere near where it needs to be, according to Beardsley. He points out that when the SARS-CoV-2 virus started spreading around the world, much of the work to counter it had already been done, including developing vaccine platforms specifically to deal with coronaviruses as well as other medicines to mitigate its effects.
Such work is still in the very early stages for a fungal pathogen outbreak, he says, and much more needs to be done to ensure health authorities have the tools to deal with such an outbreak if it does occur.
“The recent pandemic was such a good paradigm for us all to see, that basic research had already been done, so when there was a problem they could build on it,” he says.
“That work isn’t there for fungal pathogens, so there’s no way we could respond to it as quickly or effectively as we did to COVID-19.”
Fascinating answers to perplexing questions delivered to your inbox every week. Sign up to get our Explainer newsletter here.
If you'd like some expert background on an issue or a news event, drop us a line at [email protected] or [email protected]. Read more explainers here.
Most Viewed in National
Source: Read Full Article